1-in-17 women having an induced medical abortion are subsequently treated at an NHS hospital for complications arising from an incomplete abortion with retained products of conception. There have been at least 10,000 cases in England since the Government approved at-home abortion on 30 March, 2020.
Introduction.
There are 226 NHS Trusts and Foundation Trusts listed for England, of which 127 provide acute hospital services; the other 99 provide specialist services, community or mental healthcare, or ambulance services.[i]
At the start of July 2021, we sent a freedom of information request to each of the 127 NHS Trusts and Foundation Trusts which provide acute hospital services; these hospitals provide general and emergency services.
We asked the following questions:
- How many women presented to hospitals within your Trust with Retained Products of Conception (RPOC) after medically induced abortion [also known as induced miscarriage or termination of pregnancy] for the 24 months starting 01 June 2019 to 31 May 2021, and how many of them required evacuation of retained products (ERPC), [sometimes referred to as surgical management of miscarriage, SMM]. If possible, please can this information be broken down per month?
- Please tell us the threshold volume of bleeding required for coding and reporting of haemorrhage at your hospitals and how many women have been treated within your hospitals with haemorrhage after starting a medically induced abortion (also known as induced miscarriage or termination of pregnancy) for each month over a 24 month period starting 01 June 2019 to 31 May 2021.
42 Trusts, or Foundation Trusts, acknowledged our FOI request but, by the end of September 2021, failed to submit a data response. Many of the acknowledgements suggested that a reply might be delayed due to COVID-19 pressures.
85 Trusts responded with data; these represent a population of 45 million, which is about 80% of the population of England. Most Trusts include information about the population numbers served on their websites.[ii]
The Investigation team decided not to chase any late responses; we also decided not to chase any unanswered queries for clarification arising from a small number of the data responses. We decided not to chase, because of a lack of team resourcing and because we fully expected to be given, or had already received, the “COVID-19” excuse.
Many of the responses included explicit details of the search criteria, including ICD-10 codes, making it clear that data were for cases of induced medical abortion and did not include spontaneous or missed abortion. The analysis team excluded a small number of responses in which it was not possible to be certain that cases of miscarriage were not also included.
Non-negligible Risk of Failure.
It is well-known that sometimes the medical abortion treatment will fail, resulting in either a continuing pregnancy or in retained products of conception.
Ranbaxy (UK) Limited is the manufacturer of Medabon, the mifepristone/misoprostol combination treatment provided by BPAS to its pills-by-post clients. In its SmPC (summaries of product characteristics), Ranbaxy states that there is a non-negligible risk of treatment failure, as follows: [iii]
- Failures
The non-negligible risk of failure, which occurs in 4.5 to 7.8% of the cases, makes the follow-up visit mandatory in order to check that abortion is complete.
The patient should be informed that surgical treatment may be required to achieve complete abortion.
- Bleeding
The patient must be informed of the occurrence of prolonged vaginal bleeding (an average of about 13 days after mifepristone intake, up to three weeks in some women). In a few cases, heavy bleeding may require surgical evacuation of the uterus. Bleeding is not in any way a proof of termination of pregnancy as it occurs also in most cases of failure.
Linepharma, the manufacture of the mifepristone used by MSI Reproductive Choices, includes a similar warning in its SmPC: [iv]
- Failures
The non-negligible risk of failure, which occurs in up to 7.6% of the cases, makes the control visit mandatory in order to check that the expulsion is completed.
In rare case of non complete expulsion, a surgical revision may be necessary.
Though it should be noted that its use of the word ‘rare’ is incorrect, later in the same document, Linepharma notes that rare means 1-in-10,000 to 1-in-1,000; a failure rate of 7% should be noted as ‘common’. See also the SmPC for Medabon and the BNF (British National Formulary) content published by NICE.[v]
Ranbaxy and Linepharma both recognise that treatment failure will often require a surgical procedure in order to completely remove products of conception. Ranbaxy states clearly:
“Because it is important to have access to appropriate medical care if an emergency develops, the treatment procedure should only be performed where the patient has access to medical facilities equipped to provide surgical treatment for incomplete abortion”
During telemedicine consultations, women are told by BPAS and MSI Reproductive Choices that in the event of ongoing bleeding, unmanageable pain, or any concerns that something is not right, that they should go immediately to their nearest hospital emergency department.
There are three main treatments for retained products of conception: expectant management in which the medical team will adopt a wait-and-see approach; administration of an additional dose of misoprostol; and surgical evacuation (ERPC) which will be performed by the Gynae team within the hospital, rather than in the emergency department.
Data Analysis.
Trusts might not report any numbers which are less than five in any period; these are exempt under section 40(2) of the Freedom of Information Act (2000) as due to the low numbers (<5) involved it is possible that individuals may be identified. For our analysis we treated any entry of ‘<5’ as one (1).
The treatment rates for RPOC, ERPC, and haemorrhage were calculated for each of the responding Trusts in two ways and then averaged across all responses.
The rates were initially calculated using the population covered by each NHS Trust, as noted on the NHS webpages (see endnote i). We calculated the percentage of the population of England served by each Trust and then applied this percentage across the DHSC total medical abortions for England to determine a rate.[vi]
As a check and balance to the above, we also calculated the rates using the numbers of abortions reported by the DHSC for the CCG(s) and Local Authority(ies) covered by each of the Trusts.[vii]
Data submitted in the FOI responses covered the last seven months of 2019 starting 01 June; twelve months of 2020; and the first five months of 2021.
The rates in the following tables are per one hundred induced medical abortions. The first three rows are rates calculated based on population numbers and the second set of three are based on the numbers of abortions per CCG/LA.
| 2020 | 2021 | ||
| Population | RPOC | 6.2 | 6.4 |
| ERPC | 3.0 | 3.3 | |
| Haemorrhage | 2.1 | 2.3 | |
| CCG and LA | RPOC | 5.5 | 5.5 |
| ERPC | 2.7 | 3.1 | |
| Haemorrhage | 2.3 | 2.5 |
The average rates across both methods of calculation and across the whole period, are:
- 5.9% of women having an induced medical abortion are subsequently treated at an NHS hospital for complications arising from an incomplete abortion with retained products of conception.
- 3.0% of women require a surgical evacuation of retained products of conception. These are a subset of the above noted RPOC cases.
- 2.3% of women having an induced medical abortion are subsequently treated at an NHS hospital for haemorrhage.
We are aware that some women might have presented at these hospitals with RPOC saying that they were having a miscarriage, rather than admitting to using the abortion pills, in which case these rates may be understated.
We are aware that some women with complications arising from their medical abortion treatment failure may have subsequently attended one of the BPAS or MSI abortion clinics and that these will not be included in the rates above.
The rates vary from one NHS Trust to another; it may be that some facilities are more accessible and receive patients from beyond the stated locations covered and that variations may be influenced by the availability of ERPC-trained staff within each facility and/or by staff preferences for mode of follow-up treatment, some preferring e.g., not to perform the invasive surgical procedure.
As noted above, these rates are lower than those published in the SmPC for the medical abortion treatments used by BPAS and Marie Stopes: Medabon and Linepharma.
An RPOC rate of 5.9 per one hundred is consistent with Marie Stopes Australia’s published 2020 report, which indicates that the medical abortions performed by its teams fail 5.48% of the time, resulting in 4.95% incomplete abortion and 0.53% continuing pregnancy.[viii]
In March 2021, Gynuity Health Projects published a report into its study of the efficacy of telemedicine abortion services. It found that 5% of participants using the medical abortion treatment at home, needed surgical intervention to complete the procedure. Six percent of participants made unplanned visits to emergency departments or urgent care centres for reasons related to the abortion.[ix]
We can say that complications arising from the failure of the medical abortion treatment are common and should be planned for. We can expect that 1-in-17 women using the abortion pills at home, will subsequently need hospital treatment for complications arising from the medical abortion treatment failure, presenting with retained products of conception and/or haemorrhage.
The official DHSC statistics for 2020 show a 1.1% complication rate for all medical abortions and only 0.3% for medical abortions under 10-weeks. The footnote on Table 8 says: “Total complications include: haemorrhage, uterine perforation, sepsis and/or cervical tear and are those reported up to the time of discharge. Therefore complications that occur after discharge may not be recorded.” [x]
Abortion providers and the DHSC are not reporting medical abortion treatment failure as a complication, even though at least 5.9% of women using the abortion pills, need hospital treatment because of this failure. In any case, since these complications arise after discharge, the current method of reporting, using the HSA4 form, would still miss these cases even if the definition of complication were to include failed medical abortion treatment.
Less than 1-in-5 complications are being reported.
Impact of Medical Abortion Treatment Failure.
DHSC data for abortions across England show that from the start of April 2020 to the end of September 2021, 180,182 women have managed their abortions at home using either both abortion pills, or with misoprostol after being treated with mifepristone in a clinic. Each month on average, 10,000 women self-manage their medical abortion at home: 7,500 with both pills and 2,500 using misoprostol after in-clinic mifepristone.[xi]
Applying the 5.9% rate for women presenting at hospital with retained products of conception, we find more than 10,000 doing so across England since the March 2020 at-home approval for telemedicine abortion.[xii]
In England, women have been using misoprostol at home since the start of January 2019.[xiii] Since then to the end of September 2021, a total of 248,000 women will have self-managed their medical abortion at home. More than 14,000 of these women will have had treatment in an NHS hospital for RPOC, a complication arising directly from the expected failure of the medical abortion treatment.
Before the approval for use of misoprostol at home and then the approval for both pills at home, these 14,000 women may not have needed this hospital treatment. They would have had their abortion in a clinic and may not have been discharged by the abortion provider before being assessed as complete.
Sample Calculation Type 1.
Case rate of women having an induced medical abortion who subsequently need hospital treatment for retained products of conception.
Rate = ((RPOC / Pt) x Pe) / MAe x 100
Rate = ((75 / 600,000) x 56,550,000) / 110,360 x 100
Rate = 7,068.75 / 110,360 x 100
Rate = 6.41 per 100

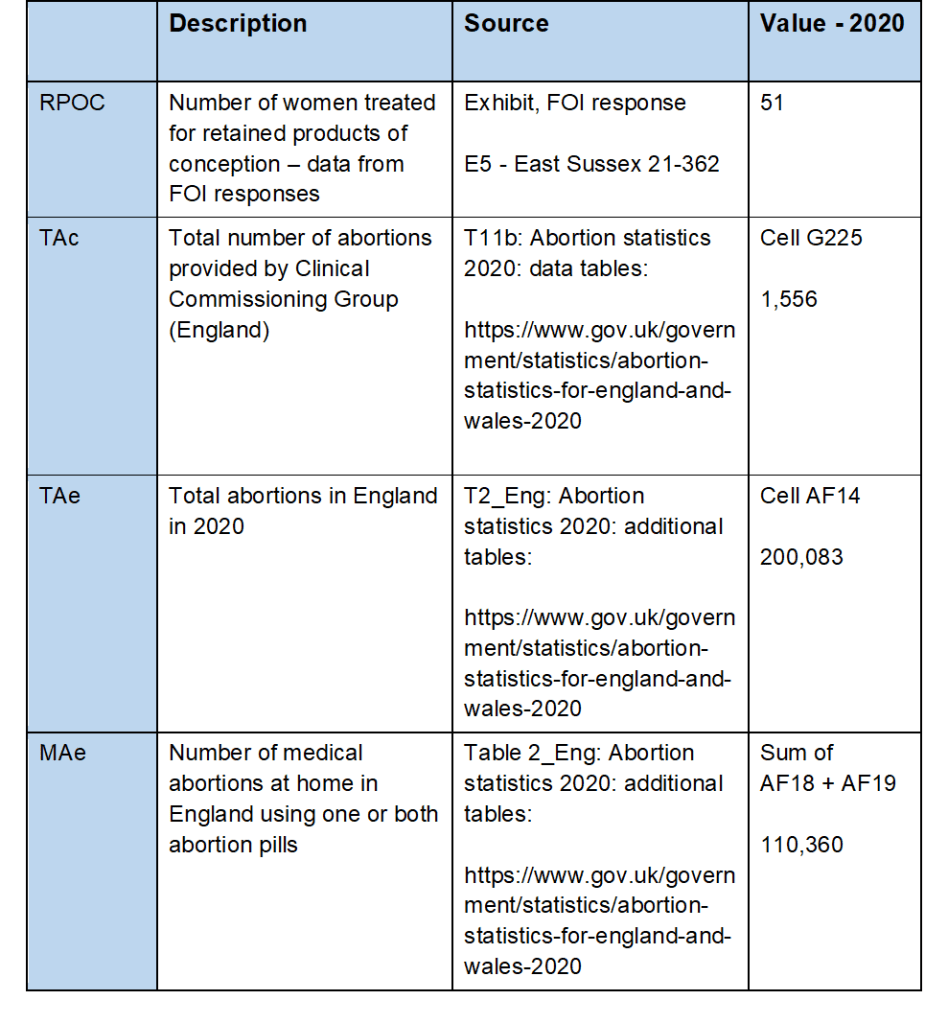
Sample Calculation Type 2 – CCG.
Case rate of women having an induced medical abortion who subsequently need hospital treatment for retained products of conception.
Rate = (RPOC / ((TAc / TAe) x MAe)) x 100
Rate = (51 / ((1,556 / 200,083) x 110,360)) x 100
Rate = (51 / 858.244) x 100
Rate = 5.94 per 100
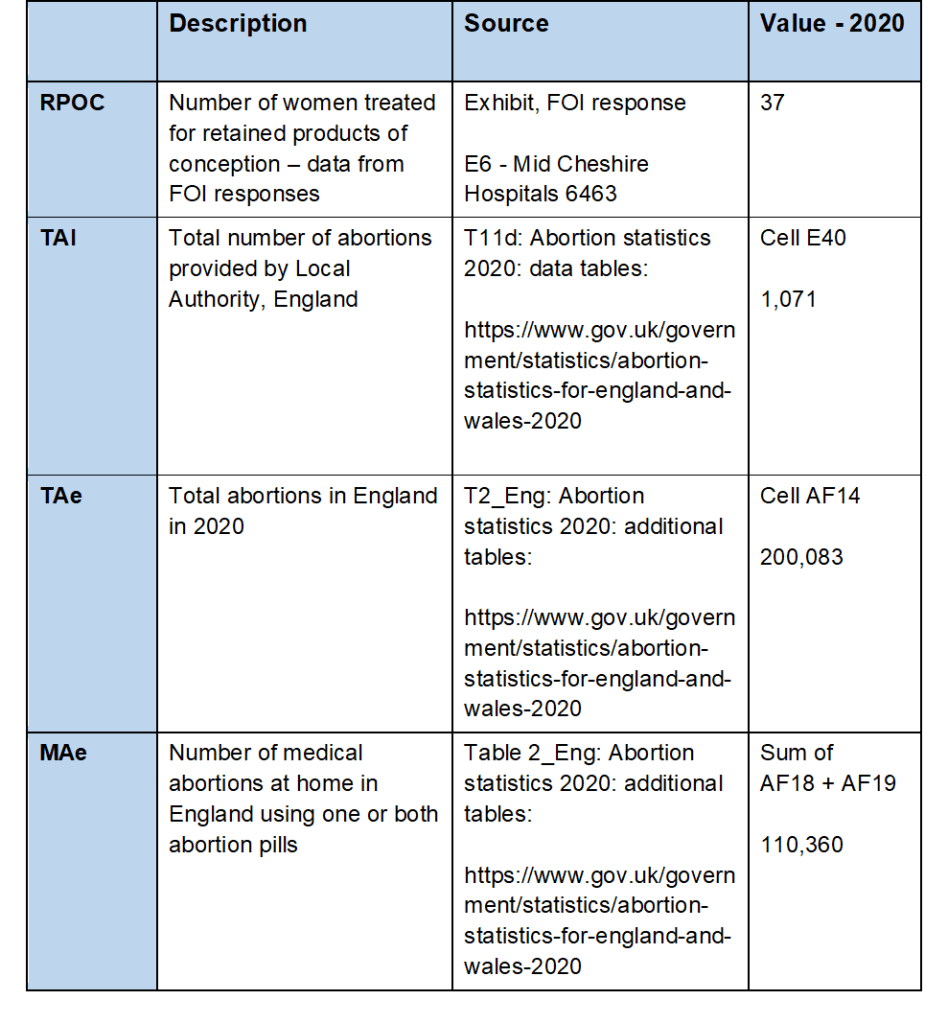
Sample Calculation Type 2 – LA.
Case rate of women having an induced medical abortion who subsequently need hospital treatment for retained products of conception.
Rate = (RPOC / ((TAl / TAe) x MAe)) x 100
Rate = (37 / ((1,071 / 200,083) x 110,360)) x 100
Rate = (37 / 590.73) x 100
Rate = 6.26 per 100
[i] NHS Trusts in England: https://www.nhs.uk/ServiceDirectories/Pages/NHSTrustListing.aspx
[ii] Ashford and St Peter’s Hospitals NHS Foundation Trust: https://www.nhs.uk/Services/Trusts/Overview/DefaultView.aspx?id=1396
[iii] Ranbaxy SmPC: https://www.medicines.org.uk/emc/product/3380/smpc
[iv] Linepharma SmPC: https://docetp.mpa.se/LMF/Mifepristone%20Linepharma%20tablet%20ENG%20SmPC_09001be6802ba369.pdf
[v] BNF, NICE: https://bnf.nice.org.uk/guidance/adverse-reactions-to-drugs.html
[vi] Abortion statistics for England and Wales: https://www.gov.uk/government/statistics/abortion-statistics-for-england-and-wales-2020
[vii] Tables 11b and 11d: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/992250/abortion-statistics-2020-data-tables_Final.ods
[viii] Marie Stopes Australia 2020 report: https://www.mariestopes.org.au/your-choices/impact-report-2020/
[ix] Gynuity Health Projects: https://gynuity.org/resources/expansion-of-a-direct-to-patient-telemedicine-abortion-service-in-the-united-states-and-experience-during-the-covid-19-pandemic
[xi] Table 2 England: https://assets.publishing.service.gov.uk/government/uploads/system/uploads/attachment_data/file/992568/All_additional_tables_Final.ods
[xii] Temporary approval of home use for both stages of early medical abortion: https://www.gov.uk/government/publications/temporary-approval-of-home-use-for-both-stages-of-early-medical-abortion–2
[xiii] Approval of home use for the second stage of early medical abortion: https://www.gov.uk/government/publications/approval-of-home-use-for-the-second-stage-of-early-medical-abortion
